Selphyl® Science
The Power of Blood
As part of the body's capacity to heal itself, platelets and other components in human blood migrate to a site of injury. Platelets are known to release a variety of factors that respond to tissue injury, where they initiate and promote healing. By concentrating platelets at the site of injury, physicians have the potential to enhance the body's natural capacity for healing.
Platelets Release Growth Factors
Cell growth, new generation and repair of blood vessels, collagen production
Growth and neogenesis of epithelial cells and vascular endothelial cells, promotion of wound healing
Promotion of angiogenesis, promotion of wound healing
Promotion of epithelial cell growth, angiogenesis, promotion of wound healing
Tissue repair, cell growth collagen production
Regulates Cell Growth
Growth of keratinocytes
Selphyl® PRP (LP-PRP)

SELPHYL® PRP tube technology was the most consistent and reproducible system tested in an independent study from Stanford (Dragoo et al.)
Leukocyte poor and red blood cell free (Dragoo et al.)
Low blood draw (9-18ccs) & short spin time (6 min)
Closed system, preloaded with sodium citrate anticoagulant; no buffering required when using sodium citrate vs. ACD-A (Mishra et al.)
Long history of safety and efficacy
4cc & 8cc PRP volumes
Up to 2-year shelf-life
The Selphyl® Process
Simple, fast, and entirely performed in your physician's office
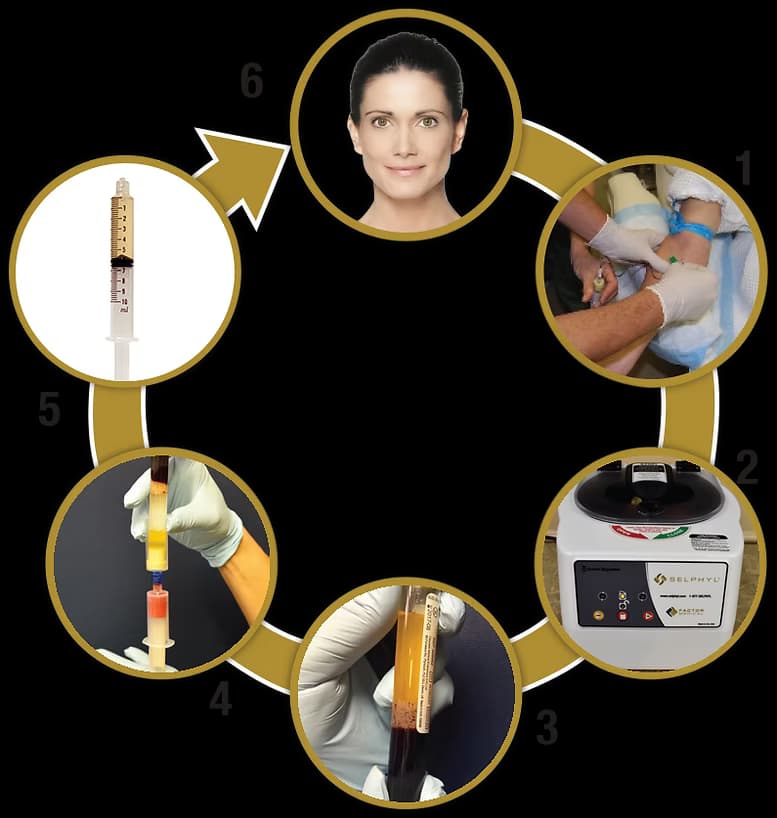
Blood Draw
During a short office visit (30 minutes), a small amount of a patient's own blood is drawn into a vacuum collection tube containing a cell separator gel.
Centrifugation
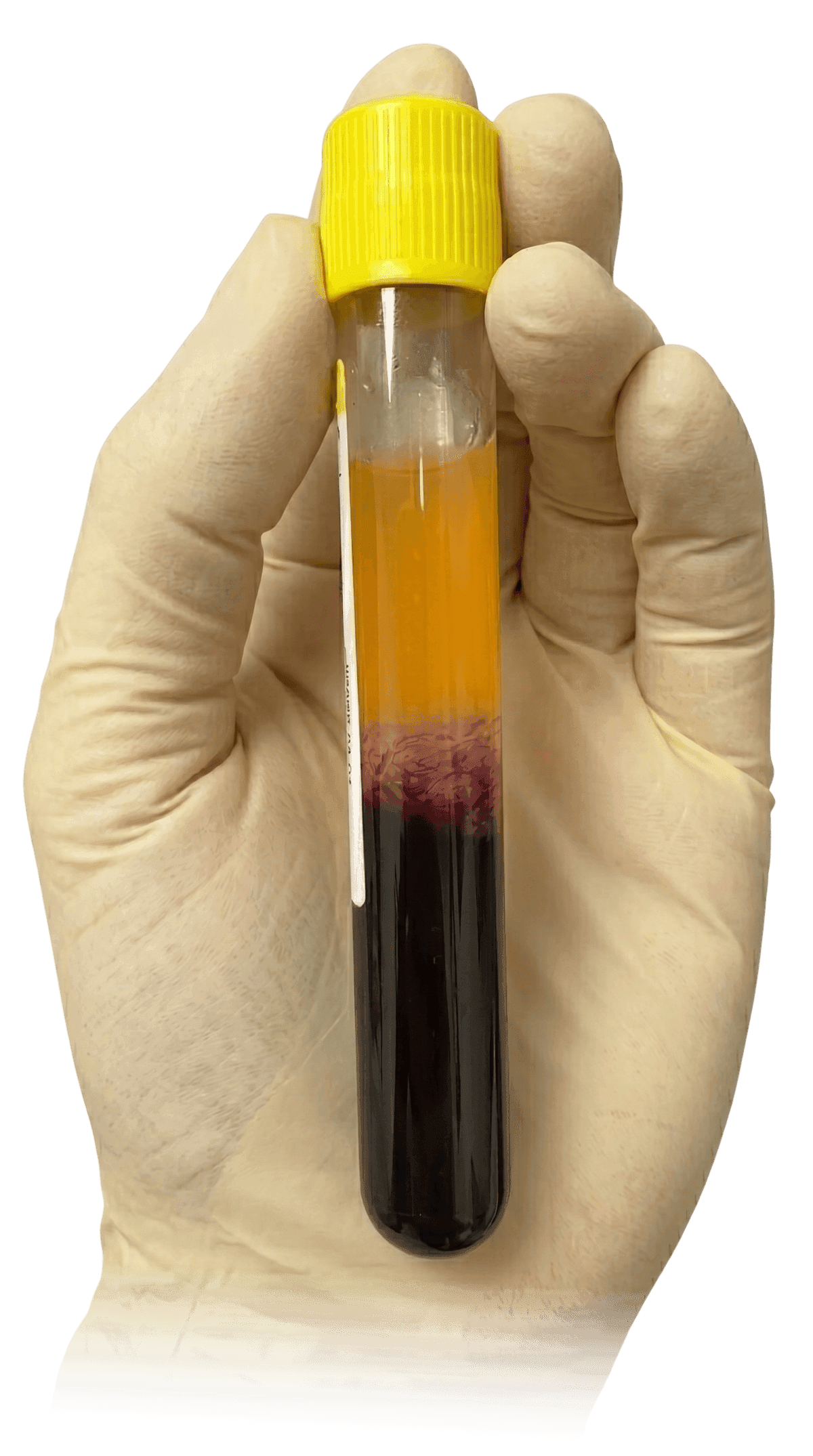
The tube is then placed into a centrifuge and spun for 6 minutes to separate the blood into a supernatant plasma/platelet suspension.
Separation
The red and white blood cells are located below the cell separator gel.
Transfer
The platelets and plasma are then transferred using a closed system, into a second vacuum tube containing a small amount of calcium chloride solution.
Activation
Fibrin polymerization begins and the PRP remains liquid for approximately 10 minutes.
Application
The patient's own PRP is ready for use as deemed appropriate by the clinical use requirements.
Is Platelet Rich Plasma Safe?
For over twenty years, PRP has been used in many different fields of medicine. Research and clinical data show PRP derived from the patient's own blood is safe, with minimal risk of adverse reactions or complications.
Because the platelets are produced from your own blood, there is no risk of rejection or disease transmission. As with any injection into the body, there is a small risk of infection, however it is very rare.
Autologous
Derived from the patient's own blood - no risk of rejection or disease transmission
No Synthetic Products
Contains no animal or synthetic products - virtually no risk of allergic reaction
Physiologic pH
Close to physiologic pH - no sodium bicarbonate buffering required before injection
The Selphyl® System
Single Kit (420910)
4cc PRP9ml blood drawDouble Kit (440910)
8cc PRP18ml blood drawCentrifuge
Reasonable centrifuge program for your practiceReferences
- Dragoo JL, Braun HJ, Durham JL, et al. Comparison of the Acute Inflammatory Response of Two Commercial Platelet-Rich Plasma Systems in Healthy Male Volunteers. Am J Sports Med. 2012;40(6):1274-1281.
- Dragoo JL, Wasterlain AS, Braun HJ, Nead KT. Platelet-Rich Plasma as a Treatment for Patellar Tendinopathy: A Double-Blind, Randomized Controlled Trial. Am J Sports Med. 2014;42(3):610-618.
- Castillo TN, Pouliot MA, Kim HJ, Rosenwasser MP. Comparison of Growth Factor and Platelet Concentration from Commercial Platelet-Rich Plasma Separation Systems. Am J Sports Med. 2011;39(2):266-271.
- Mishra A, Pavelko T. Treatment of Chronic Elbow Tendinosis with Buffered Platelet-Rich Plasma. Am J Sports Med. 2006;34(11):1774-1778.
- Fitzpatrick J, Bulsara MK, McCrory PR, Richardson MD, Zheng MH. Analysis of Platelet-Rich Plasma Extraction: Variations in Platelet and Blood Components Between 4 Common Commercial Kits. Orthop J Sports Med. 2017;5(1).
- Kon E, Buda R, Filardo G, et al. Platelet-Rich Plasma: Intra-articular Knee Injections Produced Favorable Results on Degenerative Cartilage Lesions. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):472-479.
Regulatory Status
The SELPHYL® System was cleared for marketing in 510(k) premarket notification number BK170096. Factor Medical, Inc. markets the Autologous Fibrin & Platelet System in this 510(k) under the trade names SELPHYL® and PRFM.
CAUTION: United States Federal law restricts this device to sale and distribution by or on the order of a physician, or to a clinical laboratory; and use is restricted to, by or on the order of a physician.